Public Alerts
PURPOSE: Samples seized from the Southwest border ports of entry by Customs and Border Protection (CBP) were studied by the Center for Forensic Science Research and Education (CFSRE) using advanced qualitative and quantitative testing methods at our state-of-the-art facility in Horsham, PA. This testing provides information related to understanding the health risks involved from variable drug potency and adulteration in the illicit fentanyl supply.
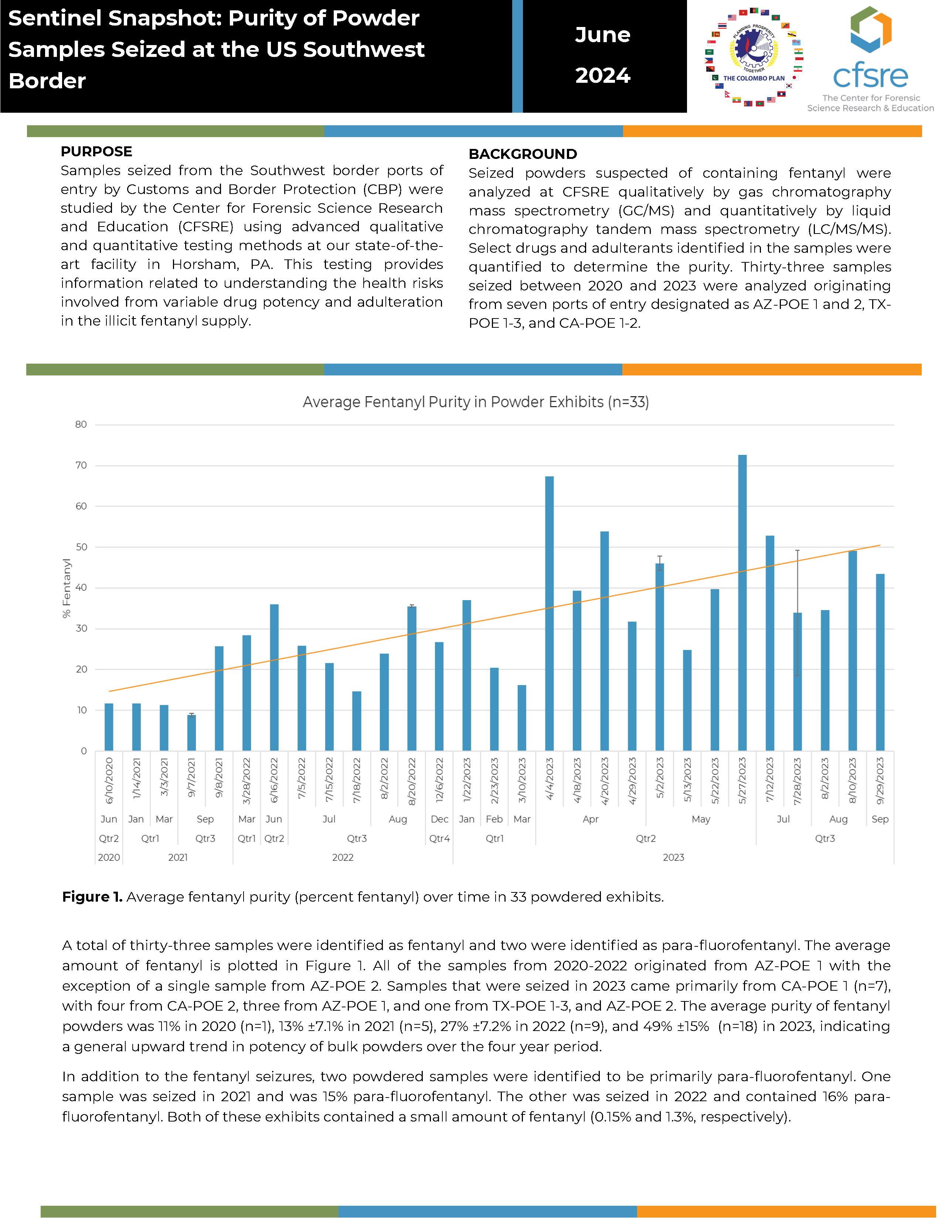
BACKGROUND: Seized powders suspected of containing fentanyl were analyzed at CFSRE qualitatively by gas chromatography mass spectrometry (GC/MS) and quantitatively by liquid chromatography tandem mass spectrometry (LC/MS/MS). Select drugs and adulterants identified in the samples were quantified to determine the purity. Thirty-three samples seized between 2020 and 2023 were analyzed originating from seven ports of entry designated as AZ-POE 1 and 2, TX-POE 1-3, and CA-POE 1-2.
PURPOSE: Samples seized from the Southwest border ports of entry by Customs and Border Protection (CBP) are submitted to the Center for Forensic Science Research and Education (CFSRE) for the purposes of qualitative and quantitative testing. The purpose of this report is to provide information on the quantitative results for tablets seized in 2020—2023.
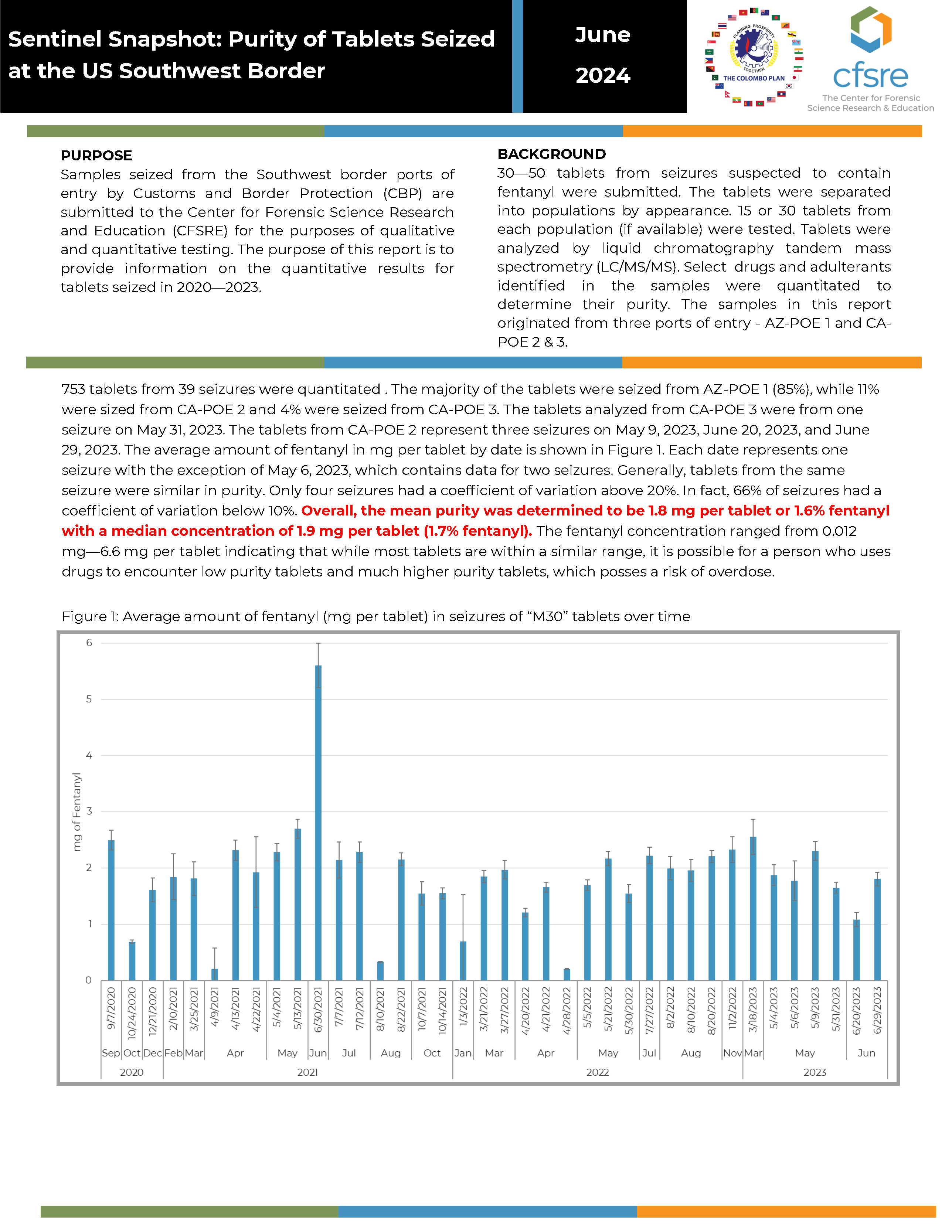
BACKGROUND: 30—50 tablets from seizures suspected to contain fentanyl were submitted. The tablets were separated into populations by appearance. 15 or 30 tablets from each population (if available) were tested. Tablets were analyzed by liquid chromatography tandem mass spectrometry (LC/MS/MS). Select drugs and adulterants identified in the samples were quantitated to determine their purity. The samples in this report originated from three ports of entry - AZ-POE 1 and CA-POE 2 & 3.
PURPOSE: Samples seized from the Southwest border ports of entry by Customs and Border Protection (CBP) were submitted to the Center for Forensic Science Research and Education (CFSRE) for the purposes of qualitative and quantitative testing. The purpose of this report is to provide information on the substances identified in counterfeit tablets seized in 2020—2023.
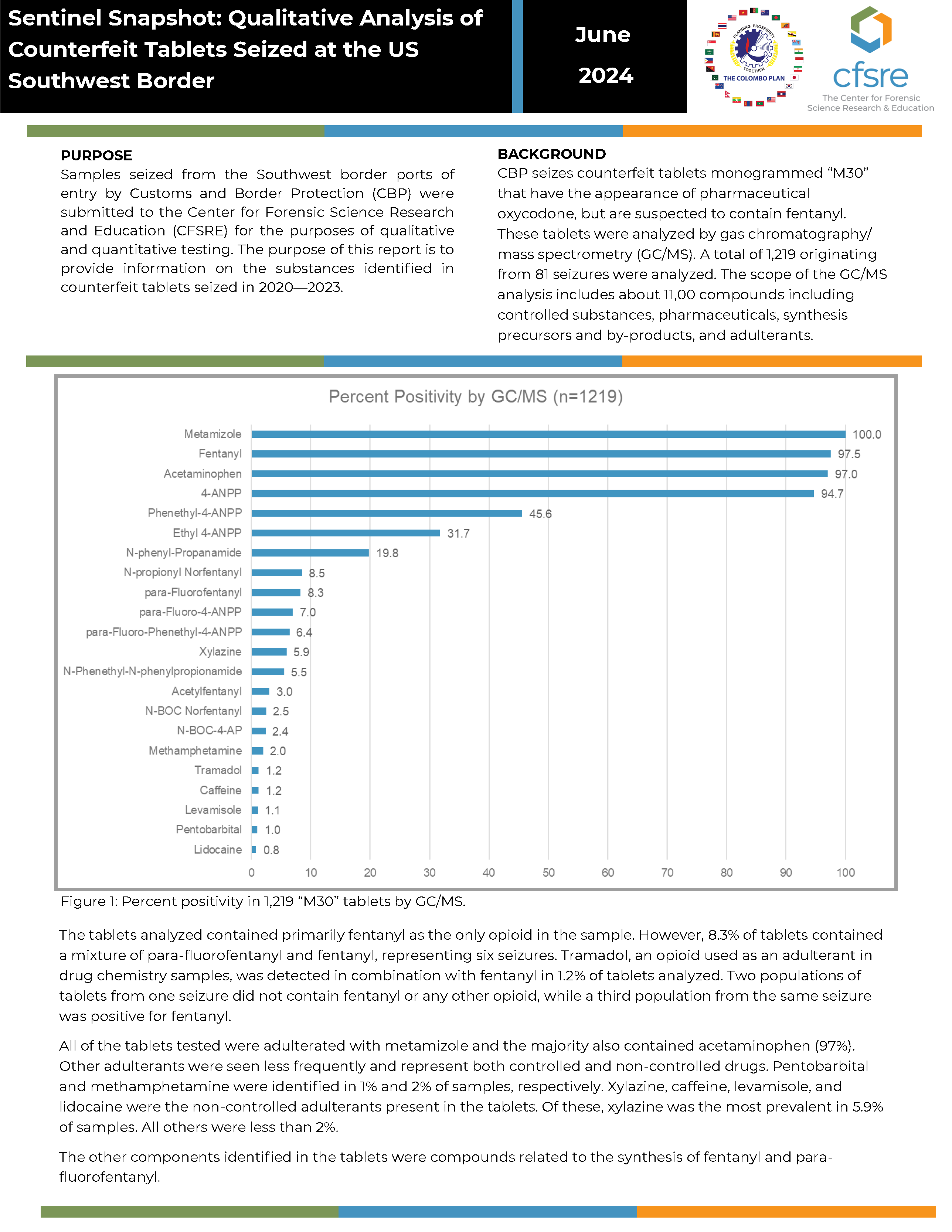
BACKGROUND: CBP seizes counterfeit tablets monogrammed “M30” that have the appearance of pharmaceutical oxycodone, but are suspected to contain fentanyl. These tablets were analyzed by gas chromatography/mass spectrometry (GC/MS). A total of 1,219 originating from 81 seizures were analyzed. The scope of the GC/MS analysis includes about 11,00 compounds including controlled substances, pharmaceuticals, synthesis precursors and by-products, and adulterants.
PURPOSE: Samples seized from the Southwest border ports of entry by Customs and Border Protection (CBP) are submitted to the Center for Forensic Science Research and Education (CFSRE) for qualitative and quantitative testing. The purpose of this report is provide information on the identification of precursors, intermediates and by-products identified in tablets and powders containing fentanyl.
BACKGROUND: Seized tablets and powders suspected of containing fentanyl are analyzed using a workflow that includes qualitative analysis by both gas chromatography mass spectrometry (GC/MS) and liquid chromatography quadrupole time-of-flight mass spectrometry (LC-QTOF-MS).
2F-2oxo-PCE is a novel synthetic hallucinogen that bears structural resemblance to ketamine and previously encountered novel dissociatives (e.g., hydroxetamine, MeO-PCE, and HO-PCE). 2F-2oxo-PCE is a positional isomer of fluorexetamine (also known as FXE or 3F-2oxo-PCE). Ketamine is regulated by the U.S. Drug Enforcement Administration (DEA) as a Schedule III drug under the Controlled Substances Act; however, neither 2F-2oxo-PCE nor fluorexetamine are currently scheduled substances. There is limited pharmacological information for 2F-2oxo-PCE; however, it is understood that 2F-2oxo-PCE is an active drug and it is hypothesized to act similarly to ketamine in vivo producing dissociative effects.
2F-2oxo-PCE was first reported in early 2022 by the Australian drug checking service CanTEST. Scientists in Canberra, Australia, first referred to the novel, unknown ketamine-like drug as “CanKet” before confirming its structure. Later that year, in December 2022, CFSRE’s NPS Discovery reported our first case involving an indiscriminate isomer of “fluorexetamine” by LC-QTOF-MS analysis only. As of May 2024, 2F-2oxo-PCE has now been identified in more than 20 drug materials and 35 toxicology specimens. More than 60% of the drug materials containing 2F-2oxo-PCE also contained opioids, primarily fentanyl. Xylazine, NPS benzodiazepines, and nitazene analogues were common co-occurrences alongside 2F-2oxo-PCE. Cases originated from at least nine states or provinces across the U.S. and Canada. In five postmortem cases, ages ranged from approx. 20 to 40 years. The toxicity of 2F-2oxo-PCE has not been formally examined but recent associations with intoxication and death lead professionals to believe that this drug has the potential to cause harm and is of high public health concern, when mixed with other drugs or when consumed alone at high doses.
Purpose: The objective of this announcement is to notify public health, harm reduction, first responders, clinicians, medical examiners and coroners, forensic and clinical laboratories, and all other related communities about new information surrounding the emergent adulterant medetomidine (also referred to as dexmedetomidine).
Background: Medetomidine is an alpha-2 agonist, belonging to the same family of drugs as xylazine and clonidine. Medetomidine is synthetically manufactured and exists in two enantiomeric forms: dexmedetomidine and levomedetomidine, the former being active and potent. Dexmedetomidine is approved for use in humans and is administered in hospital, while differing forms of medetomidine are available for use in veterinary medicine. The effects of medetomidine can include sedation, analgesia, muscle relaxation, anxiolysis, bradycardia, hypotension, hyperglycemia, and hallucinations. Duration of action is noted to be longer for medetomidine relative to xylazine.
Summary: Medetomidine is the latest CNS depressant to appear as an adulterant alongside fentanyl in the recreational drug supply. Recent mass overdose outbreaks in Philadelphia, Chicago, and elsewhere have all been associated with fentanyl or heroin drug products containing medetomidine, as well xylazine and/or other substances. In cases where medetomidine ingestion is suspected or confirmed, severe adverse effects have been noted, including heightened sedation and profound bradycardia. In December 2023, the CFSRE and the Colombo Plan issued a Toxic Adulterant Alert for medetomidine following its emergence in the recreational drug supply.
para-Fluorofentanyl (pFF) has been reported as an adulterant in the illicit drug supply in recent years, particularly with the rise in illicit fentanyl. pFF is a fentanyl analogue, and is considered a narcotic analgesic. In laboratory studies, pFF is approximately three times less potent compared to fentanyl, however, concentrations in drug deaths suggest that deaths can occur with concentrations similar to those involving fentanyl. pFF is believed to be added to fentanyl to increase total drug volume for distribution, to dilute the drug being cut and to enhance the pharmacological effect. Its increase in prevalence may also be a result of transition by illicit manufacturers to more readily available precursors. Its adverse effects include sedation, respiratory depression, coma, and potentially death. In a study at the Fredric Rieders Family Foundation (FRFF) supported by the Colombo Plan on the presence of toxic adulterants in heroin/fentanyl, methamphetamine and cocaine seized drugs case in the United States (n=2,027), pFF was found in approximately 3% percent of the total exhibits and almost always found together with fentanyl (93% of the time). CNS stimulants, methamphetamine and/or cocaine were identified in 19 of the 60 cases, generally in addition to other opioids and adulterants. Of the 60 cases positive for pFF, there were four cases where fentanyl was not detected. One case was pFF only, one was pFF and tramadol, one was pFF and a fentanyl synthesis precursor, and the last case contained pFF and the novel opioid, N-Pyrrolidino Etonitazene (etonitazepyne).
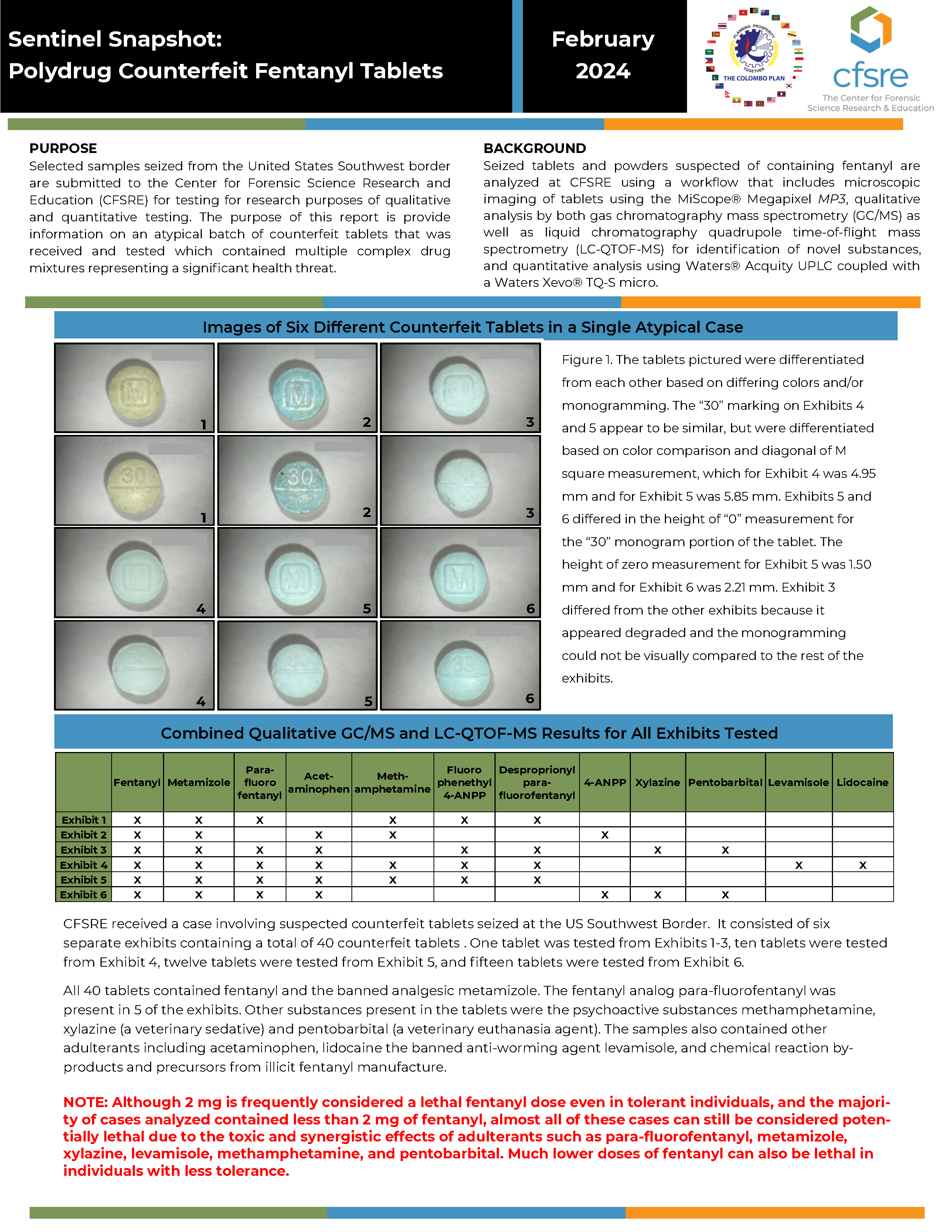
Selected samples seized from the United States Southwest border are submitted to the Center for Forensic Science Research and Education (CFSRE) for testing for research purposes of qualitative and quantitative testing. The purpose of this report is provide information on an atypical batch of counterfeit tablets that was received and tested which contained multiple complex drug mixtures representing a significant health threat.
Seized tablets and powders suspected of containing fentanyl are analyzed at CFSRE using a workflow that includes microscopic imaging of tablets using the MiScope® Megapixel MP3, qualitative analysis by both gas chromatography mass spectrometry (GC/MS) as well as liquid chromatography quadrupole time-of-flight mass spectrometry (LC-QTOF-MS) for identification of novel substances, and quantitative analysis using Waters® Acquity UPLC coupled with a Waters Xevo® TQ-S micro.
DOWNLOAD THE ALERT
Tianeptine, popularly known as “gas station heroin” has been encountered in various forms including bulk powder and counterfeit pills mimicking hydrocodone and oxycodone. Tianeptine has been sold as “ZaZa”,. “Tiana”, “Neptune’s Fix” and other brand names. Poison Control Center cases involving tianeptine exposure increased nationwide, from 11 total cases between 2000 and 2013 to 151 cases in 2020. As an antidepressant, tianeptine is prescribed to treat depression and anxiety in some European, Asian, and South American countries, but it is not approved for medical use in the United States. Tianeptine-containing products are marketed as dietary supplements or as “smart drugs” that allegedly enhance cognitive function. Tianeptine induces euphoria at high doses via activation of mu-opioid and dopaminergic receptors. It is also alleged to be useful to manage opioid consumption, with claims that it reduces the effects of opioid withdrawal and craving. Unregulated tianeptine is at an increased risk of contamination with adulterants, which may cause additional or unexpected side effects. Tianeptine is not currently controlled under the Controlled Substances Act, but has been scheduled in several states.
DOWNLOAD THE ALERT- Fentanyl misuse or abuse typically involves injection or smoking of illicitly manufactured fentanyl (IMF). Illicit fentanyl is most commonly sold on the street as counterfeit tablets or as powders in folded glassine papers.
- Recently, however, diverted injectable solutions of pharmaceutical fentanyl have been reported in countries where IMF has not yet become established, such as Nigeria, Colombia and El Salvador (see details over); in addition to Argentina (500 vials), Brazil (72 vials), and Costa Rica (25 vials). Furthermore, there are reports of fentanyl injectable solutions being stolen or diverted from hospitals, clinics and medical supply houses in Panama (19,000 vials), the United States, and Europe.